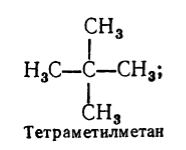
Alkanes :
Alkanes are saturated hydrocarbons, in the molecules of which all atoms are connected by single bonds. Formula -
Physical Properties :
- Melting and boiling points increase with molecular weight and main carbon chain length
- Under normal conditions, unbranched alkanes from CH 4 to C 4 H 10 are gases; from C 5 H 12 to C 13 H 28 - liquids; after C 14 H 30 - solids.
- The melting and boiling points decrease from less branched to more branched. So, for example, at 20 °C, n-pentane is a liquid, and neopentane is a gas.
Chemical properties:
· Halogenation
this is one of the substitution reactions. The least hydrogenated carbon atom is halogenated first (tertiary atom, then secondary, primary atoms are halogenated last). Halogenation of alkanes takes place in stages - no more than one hydrogen atom is replaced in one stage:
- CH 4 + Cl 2 → CH 3 Cl + HCl (chloromethane)
- CH 3 Cl + Cl 2 → CH 2 Cl 2 + HCl (dichloromethane)
- CH 2 Cl 2 + Cl 2 → CHCl 3 + HCl (trichloromethane)
- CHCl 3 + Cl 2 → CCl 4 + HCl (tetrachloromethane).
Under the action of light, the chlorine molecule decomposes into radicals, then they attack the alkane molecules, taking a hydrogen atom from them, as a result of which methyl radicals CH 3 are formed, which collide with chlorine molecules, destroying them and forming new radicals.
· Combustion
The main chemical property of saturated hydrocarbons, which determine their use as a fuel, is the combustion reaction. Example:
CH 4 + 2O 2 → CO 2 + 2H 2 O + Q
In the event of a lack of oxygen, instead of carbon dioxide, carbon monoxide or coal is obtained (depending on the oxygen concentration).
In general, the combustion reaction of alkanes can be written as follows:
With n H 2 n +2 +(1,5n+0.5)O 2 \u003d n CO 2 + ( n+1) H 2 O
· Decomposition
Decomposition reactions occur only under the influence of high temperatures. An increase in temperature leads to the breaking of the carbon bond and the formation of free radicals.
Examples:
CH 4 → C + 2H 2 (t > 1000 °C)
C 2 H 6 → 2C + 3H 2
Alkenes :
Alkenes are unsaturated hydrocarbons containing in the molecule, in addition to single bonds, one double carbon-carbon bond. The formula is C n H 2n
The belonging of a hydrocarbon to the class of alkenes is reflected by the generic suffix -ene in its name.
Physical Properties :
- The melting and boiling points of alkenes (simplified) increase with molecular weight and length of the main carbon chain.
- Under normal conditions, alkenes from C 2 H 4 to C 4 H 8 are gases; from C 5 H 10 to C 17 H 34 - liquids, after C 18 H 36 - solids. Alkenes are insoluble in water, but readily soluble in organic solvents.
Chemical properties :
· Dehydration is the process of splitting a water molecule from an organic compound molecule.
· Polymerization- this is a chemical process of combining many initial molecules of a low molecular weight substance into large polymer molecules.
Polymer is a high molecular weight compound, the molecules of which consist of many identical structural units.
Alkadienes :
Alkadienes are unsaturated hydrocarbons containing in the molecule, in addition to single bonds, two double carbon-carbon bonds. The formula is
. Dienes are structural isomers of alkynes.Physical Properties :
Butadiene is a gas (tboiling −4.5 °C), isoprene is a liquid boiling at 34 °C, dimethylbutadiene is a liquid boiling at 70 °C. Isoprene and other diene hydrocarbons are able to polymerize into rubber. Natural rubber in its purified state is a polymer with the general formula (C5H8)n and is obtained from the latex of certain tropical plants.
Rubber is highly soluble in benzene, gasoline, carbon disulfide. At low temperature it becomes brittle, when heated it becomes sticky. To improve the mechanical and chemical properties of rubber, it is converted into rubber by vulcanization. To obtain rubber products, they are first molded from a mixture of rubber with sulfur, as well as with fillers: soot, chalk, clay and some organic compounds that serve to accelerate vulcanization. Then the products are heated - hot vulcanization. During vulcanization, sulfur chemically bonds with rubber. In addition, in vulcanized rubber, sulfur is contained in a free state in the form of tiny particles.
Diene hydrocarbons are easily polymerized. The polymerization reaction of diene hydrocarbons underlies the synthesis of rubber. Enter into addition reactions (hydrogenation, halogenation, hydrohalogenation):
H 2 C \u003d CH-CH \u003d CH 2 + H 2 -> H 3 C-CH \u003d CH-CH 3
Alkynes :
Alkynes are unsaturated hydrocarbons whose molecules contain, in addition to single bonds, one triple carbon-carbon bond. Formula-C n H 2n-2
Physical Properties :
Alkynes are similar in physical properties to the corresponding alkenes. Lower (up to C 4) - gases without color and odor, having higher boiling points than their counterparts in alkenes.
Alkynes are poorly soluble in water, better in organic solvents.
Chemical properties :
halogenation reactions
Alkynes are capable of adding one or two halogen molecules to form the corresponding halogen derivatives:
Hydration
In the presence of mercury salts, alkynes add water to form acetaldehyde (for acetylene) or ketone (for other alkynes)

DEFINITION
Alkanes saturated hydrocarbons are called, the molecules of which consist of carbon and hydrogen atoms, linked to each other only by σ-bonds.
Under normal conditions (at 25 o C and atmospheric pressure), the first four members of the homologous series of alkanes (C 1 - C 4) are gases. Normal alkanes from pentane to heptadecane (C 5 - C 17) are liquids, starting from C 18 and above are solids. As the relative molecular weight increases, the boiling and melting points of alkanes increase. With the same number of carbon atoms in a molecule, branched alkanes have lower boiling points than normal alkanes. The structure of the alkanes molecule using methane as an example is shown in fig. one.
Rice. 1. The structure of the methane molecule.
Alkanes are practically insoluble in water, since their molecules are of low polarity and do not interact with water molecules. Liquid alkanes mix easily with each other. They dissolve well in non-polar organic solvents such as benzene, carbon tetrachloride, diethyl ether, etc.
Obtaining alkanes
The main sources of various saturated hydrocarbons containing up to 40 carbon atoms are oil and natural gas. Alkanes with a small number of carbon atoms (1 - 10) can be isolated by fractional distillation of natural gas or gasoline fraction of oil.
There are industrial (I) and laboratory (II) methods for obtaining alkanes.
C + H 2 → CH 4 (kat = Ni, t 0);
CO + 3H 2 → CH 4 + H 2 O (kat \u003d Ni, t 0 \u003d 200 - 300);
CO 2 + 4H 2 → CH 4 + 2H 2 O (kat, t 0).
— hydrogenation of unsaturated hydrocarbons
CH 3 -CH \u003d CH 2 + H 2 →CH 3 -CH 2 -CH 3 (kat \u003d Ni, t 0);
— recovery of haloalkanes
C 2 H 5 I + HI → C 2 H 6 + I 2 (t 0);
- alkaline melting reactions of salts of monobasic organic acids
C 2 H 5 -COONa + NaOH → C 2 H 6 + Na 2 CO 3 (t 0);
- interaction of haloalkanes with metallic sodium (Wurtz reaction)
2C 2 H 5 Br + 2Na → CH 3 -CH 2 -CH 2 -CH 3 + 2NaBr;
– electrolysis of salts of monobasic organic acids
2C 2 H 5 COONa + 2H 2 O → H 2 + 2NaOH + C 4 H 10 + 2CO 2;
K (-): 2H 2 O + 2e → H 2 + 2OH -;
A (+): 2C 2 H 5 COO - -2e → 2C 2 H 5 COO + → 2C 2 H 5 + + 2CO 2.
Chemical properties of alkanes
Alkanes are among the least reactive organic compounds, which is explained by their structure.
Alkanes under normal conditions do not react with concentrated acids, molten and concentrated alkalis, alkali metals, halogens (except fluorine), potassium permanganate and potassium dichromate in an acidic environment.
For alkanes, reactions proceeding according to the radical mechanism are most characteristic. The homolytic cleavage of C-H and C-C bonds is energetically more favorable than their heterolytic cleavage.
Radical substitution reactions proceed most easily at the tertiary carbon atom, more easily at the secondary carbon atom, and lastly at the primary carbon atom.
All chemical transformations of alkanes proceed with splitting:
1) C-H bonds
- halogenation (S R)
CH 4 + Cl 2 → CH 3 Cl + HCl ( hv);
CH 3 -CH 2 -CH 3 + Br 2 → CH 3 -CHBr-CH 3 + HBr ( hv).
- nitration (S R)
CH 3 -C (CH 3) H-CH 3 + HONO 2 (dilute) → CH 3 -C (NO 2) H-CH 3 + H 2 O (t 0).
– sulfochlorination (S R)
R-H + SO 2 + Cl 2 → RSO 2 Cl + HCl ( hv).
– dehydrogenation
CH 3 -CH 3 → CH 2 \u003d CH 2 + H 2 (kat \u003d Ni, t 0).
— dehydrocyclization
CH 3 (CH 2) 4 CH 3 → C 6 H 6 + 4H 2 (kat = Cr 2 O 3, t 0).
2) C-H and C-C bonds
- isomerization (intramolecular rearrangement)
CH 3 -CH 2 -CH 2 -CH 3 →CH 3 -C (CH 3) H-CH 3 (kat \u003d AlCl 3, t 0).
- oxidation
2CH 3 -CH 2 -CH 2 -CH 3 + 5O 2 → 4CH 3 COOH + 2H 2 O (t 0, p);
C n H 2n + 2 + (1.5n + 0.5) O 2 → nCO 2 + (n + 1) H 2 O (t 0).
Application of alkanes
Alkanes have found application in various industries. Let us consider in more detail, using the example of some representatives of the homologous series, as well as mixtures of alkanes.
Methane is the raw material basis of the most important chemical industrial processes for producing carbon and hydrogen, acetylene, oxygen-containing organic compounds - alcohols, aldehydes, acids. Propane is used as an automotive fuel. Butane is used to produce butadiene, which is a raw material for the production of synthetic rubber.
A mixture of liquid and solid alkanes up to C 25, called vaseline, is used in medicine as the basis for ointments. A mixture of solid alkanes C 18 - C 25 (paraffin) is used to impregnate various materials (paper, fabrics, wood) to give them hydrophobic properties, i.e. water impermeability. In medicine, it is used for physiotherapeutic procedures (paraffin treatment).
Examples of problem solving
EXAMPLE 1
| Exercise | When chlorinating methane, 1.54 g of the compound was obtained, the vapor density in air of which is 5.31. Calculate the mass of manganese dioxide MnO 2 that will be required to produce chlorine if the ratio of the volumes of methane and chlorine introduced into the reaction is 1:2. |
| Decision | The ratio of the mass of a given gas to the mass of another gas taken in the same volume, at the same temperature and the same pressure, is called the relative density of the first gas over the second. This value shows how many times the first gas is heavier or lighter than the second gas. The relative molecular weight of air is taken equal to 29 (taking into account the content of nitrogen, oxygen and other gases in the air). It should be noted that the concept of "relative molecular weight of air" is used conditionally, since air is a mixture of gases. Let's find the molar mass of the gas formed during the chlorination of methane: M gas \u003d 29 × D air (gas) \u003d 29 × 5.31 \u003d 154 g / mol. This is carbon tetrachloride - CCl 4 . We write the reaction equation and arrange the stoichiometric coefficients: CH 4 + 4Cl 2 \u003d CCl 4 + 4HCl. Calculate the amount of carbon tetrachloride substance: n(CCl 4) = m(CCl 4) / M(CCl 4); n (CCl 4) \u003d 1.54 / 154 \u003d 0.01 mol. According to the reaction equation n (CCl 4) : n (CH 4) = 1: 1, then n (CH 4) \u003d n (CCl 4) \u003d 0.01 mol. Then, the amount of chlorine substance should be equal to n(Cl 2) = 2 × 4 n(CH 4), i.e. n(Cl 2) \u003d 8 × 0.01 \u003d 0.08 mol. We write the reaction equation for the production of chlorine: MnO 2 + 4HCl \u003d MnCl 2 + Cl 2 + 2H 2 O. The number of moles of manganese dioxide is 0.08 moles, because n (Cl 2) : n (MnO 2) = 1: 1. Find the mass of manganese dioxide: m (MnO 2) \u003d n (MnO 2) × M (MnO 2); M (MnO 2) \u003d Ar (Mn) + 2 × Ar (O) \u003d 55 + 2 × 16 \u003d 87 g / mol; m (MnO 2) \u003d 0.08 × 87 \u003d 10.4 g. |
| Answer | The mass of manganese dioxide is 10.4 g. |
EXAMPLE 2
| Exercise | Set the molecular formula of trichloroalkane, the mass fraction of chlorine in which is 72.20%. Make up the structural formulas of all possible isomers and give the names of substances according to the substitutional IUPAC nomenclature. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Answer | Let's write the general formula of trichloroalkene: C n H 2 n -1 Cl 3 . According to the formula ω(Cl) = 3×Ar(Cl) / Mr(C n H 2 n -1 Cl 3) × 100% calculate the molecular weight of trichloroalkane: Mr(C n H 2 n -1 Cl 3) = 3 × 35.5 / 72.20 × 100% = 147.5. Let's find the value of n: 12n + 2n - 1 + 35.5x3 = 147.5; Therefore, the formula of trichloroalkane is C 3 H 5 Cl 3. Let us compose the structural formulas of the isomers: 1,2,3-trichloropropane (1), 1,1,2-trichloropropane (2), 1,1,3-trichloropropane (3), 1,1,1-trichloropropane (4) and 1 ,2,2-trichloropropane (5). CH 2 Cl-CHCl-CH 2 Cl (1); CHCl 2 -CHCl-CH 3 (2); CHCl 2 -CH 2 -CH 2 Cl (3); CCl 3 -CH 2 -CH 3 (4); Alkanes (methane and its homologues) have the general formula C n H2 n+2. The first four hydrocarbons are called methane, ethane, propane, butane. The names of the higher members of this series consist of the root - the Greek numeral and the suffix -an. The names of alkanes form the basis of the IUPAC nomenclature.Rules for systematic nomenclature:
The main circuit is selected based on the following criteria in sequence:
The main chain is numbered from one end to the other in Arabic numerals. Each substituent receives the number of the carbon atom of the main chain to which it is attached. The numbering sequence is chosen in such a way that the sum of the numbers of substituents (locants) is the smallest. This rule also applies to the numbering of monocyclic compounds.
All hydrocarbon side groups are considered as monovalent (singly bonded) radicals. If the side radical itself contains side chains, then an additional main chain is selected in it according to the above rules, which is numbered starting from the carbon atom attached to the main chain.
The name of the compound begins with a list of substituents, indicating their names in alphabetical order. The name of each substituent is preceded by its number in the main chain. The presence of several substituents is indicated by prefix-numerators: di-, tri-, tetra-, etc. After that, the hydrocarbon corresponding to the main chain is called. In table. 12.1 shows the names of the first five hydrocarbons, their radicals, possible isomers and their corresponding formulas. The names of radicals end with the suffix -yl.
Example. Name all isomers of hexane. Example. Name the alkane of the following structure In this example, of two twelve-atomic chains, the one in which the sum of the numbers is the smallest is chosen (rule 2). Using the names of branched radicals given in Table. 12.2,
the name of this alkane is somewhat simplified: 10-tert-butyl-2,2-(dimethyl)-7-propyl-4-isopropyl-3-ethyl dodecane. When the hydrocarbon chain is closed in a cycle with the loss of two hydrogen atoms, monocycloalkanes are formed with the general formula C n H2 n. Cyclization starts from C 3, names are formed from C n prefixed with cyclo: polycyclic alkanes. Their names are formed by the prefix bicyclo-, tricyclo-, etc. Bicyclic and tricyclic compounds contain two and three cycles in the molecule, respectively; to describe their structure, in square brackets indicate in decreasing order the number of carbon atoms in each of the chains connecting the nodal atoms ; under the formula the name of the atom: This tricyclic hydrocarbon is commonly referred to as adamantane (from the Czech adamant, diamond) because it is a combination of three fused cyclohexane rings in a form that results in a diamond-like arrangement of carbon atoms in the crystal lattice. Cyclic hydrocarbons with one common carbon atom are called spiranes, for example, spiro-5,5-undecane: Planar cyclic molecules are unstable, so various conformational isomers are formed. Unlike configurational isomers (the spatial arrangement of atoms in a molecule without regard to orientation), conformational isomers differ from each other only by the rotation of atoms or radicals around formally simple bonds while maintaining the configuration of molecules. The energy of formation of a stable conformer is called conformational. Conformers are in dynamic equilibrium and are converted into each other through unstable forms. The instability of planar cycles is caused by a significant deformation of bond angles. While maintaining the tetrahedral bond angles for cyclohexane C 6H 12, two stable conformations are possible: in the form of a chair (a) and in the form of a bath (b):
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||